|
1/1/2024 0 Comments Moderna embryonic stem cells Regeneron spokesperson Alexandra Bowie previously told MIT Technology Review that "the 293T cell lines available today are not considered fetal tissue, and we did not otherwise use fetal tissue. Like many other science-focused organizations, we do use these tools in a responsible and limited manner for certain early research." They added: "We did not use human stem cells or human embryonic stem cells in our COVID-19 program. 
That gives them the potential to treat a dizzying array of conditions, from Parkinson’s disease. "It's commercially available and very commonly used by research labs and have been involved in the development of important vaccines and therapeutics for conditions such as hemophilias A/B & Type 2 diabetes." In an early phase of development, two of the coronavirus shots the mRNA vaccines from Pfizer/BioNTech and Moderna were tested in cell lines that were long ago made from an aborted fetus. In a feat of biological alchemy, embryonic stem cells can turn into any type of cell in the body. "HEK293T is a 40+ year old 'immortalized' cell line that was originally derived from embryonic kidney cells in the 1970s. 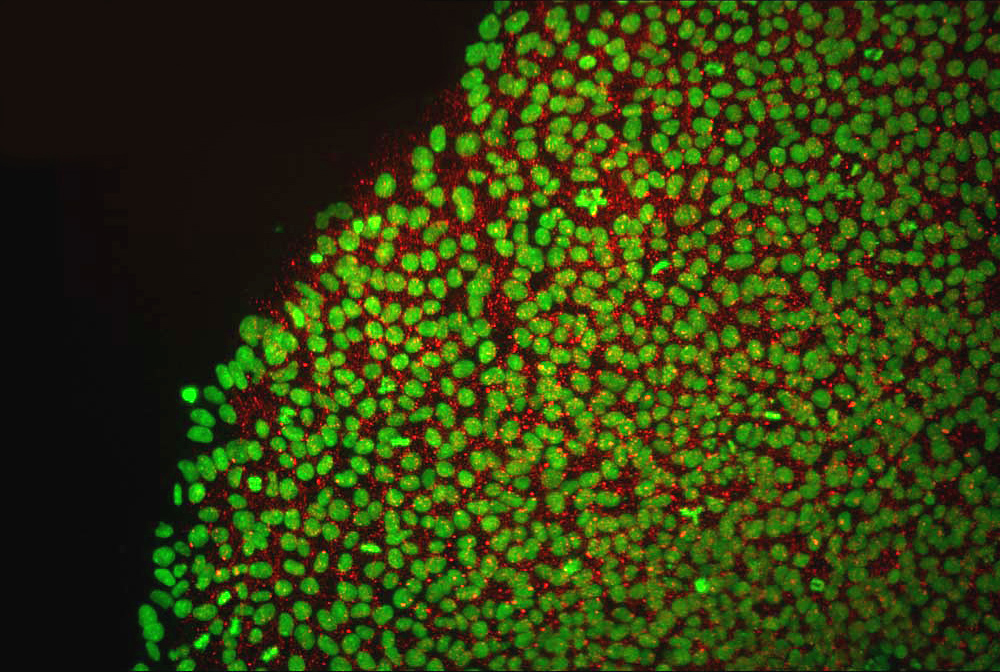
"Of the many experiments, one involved using the HEK293T cell line to help us select potent virus-neutralizing antibodies," they said. The cells are cloned and those used in recent times are not from the original fetal tissue, though that is where they originated.Ī spokesperson for Regeneron confirmed to Newsweek one experiment in testing the treatment used the HEK293T cell line. HEK293 cells were originally "derived in 1973 from the kidney of an aborted human embryo of unknown parenthood," according to a 2014 article in the science journal Nature. These cells are a variant of HEK293 cells. They were donated specially for this project with the informed consent of donors. These embryos were produced for clinical purposes, but were no longer wanted for implantation by the couples who donated them. Here are the 10 top stem cell companies on the. In the development of the Regeneron treatment, the HEK293T cell line was used in an experiment during the testing process. Human embryonic stem cells are derived from in vitro fertilized embryos less than a week old. The global stem cell therapy market is expected to experience significant growth over the next few years. It is not deemed a substitute for vaccination. The breakthrough could help research into genetic disorders and in understanding. Food and Drug Administration ( FDA) authorized the treatment for "emergency use as post-exposure prophylaxis (prevention) for COVID-19 in adults and pediatric individuals (12 years of age and older weighing at least 40 kg) who are at high risk for progression to severe COVID-19, including hospitalization or death." Scientists have created a stem cell-derived model of the human embryo in the lab by reprogramming human stem cells. Dr Shoukhrat Mitalipov said: 'A thorough examination of the stem cells derived through this technique demonstrated their ability to convert just like normal embryonic stem cells, into. These antibodies are administered to people to treat COVID-19. While Moderna and Pfizer used fetal cells for testing their vaccine after it was already produced, J&J used fetal cells as tiny 'factories' that produced the active ingredient in its vaccine. Regeneron's monoclonal antibody treatment, REGEN-COV, is a mixture of two separate monoclonal antibodies-casirivimab and imdevimab. The hypocrisy never stops.- Kaivan Shroff September 1, 2021 The COVID-19 vaccine (J&J/Janssen) is made using fetal retinal cells. 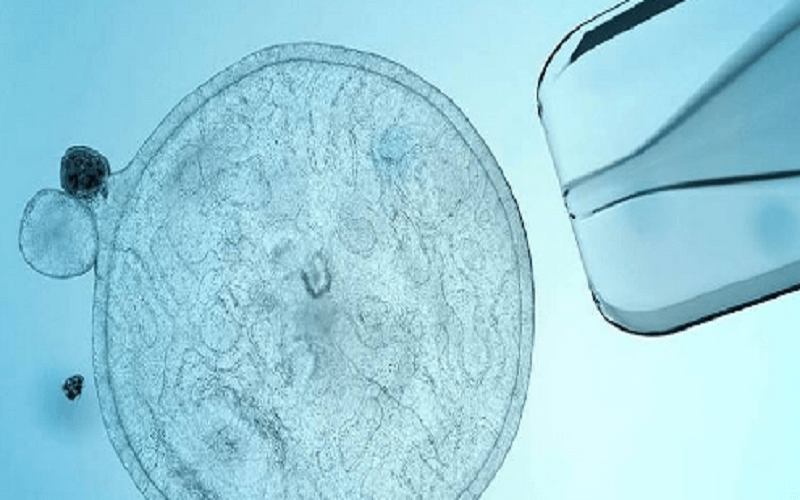
However, with expected future surges in infections due to new variants, employers now have an option for responding to some employees’ religious objections to their vaccination mandates.Just days ago Texas Governor Greg Abbott took Regeneron for COVID - a drug tested on cells derived from fetal tissue. Outside of the healthcare industry, many employers have dropped COVID-19 vaccination mandates. Of course, many workers requested religious exemptions based on their objection to the use of any vaccinations. Presumably, employees who were granted exemptions based on this reasoning would not have religious objections to obtaining the Novavax vaccine. Many employers that mandated COVID-19 vaccinations for employees received requests for religious exemptions based on the fact that stem-cell lines used to develop the vaccines may have been originally obtained from aborted fetuses. This approval raises interesting questions and options for employers and employees. Unlike any of the other approved vaccinations, the Novavax shot was not developed through the use of fetal stem-cell lines. Unlike the Pfizer and Moderna products, this vaccine does not use messenger RNA technology. Last week, the Food and Drug Administration (FDA) authorized a fourth coronavirus vaccination, approving a new shot developed by Novavax. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
